The ultimate battery made from air is expected to appear in the near future. The battery does not require traditional electrodes and is 1/5 the weight of existing lithium batteries. "Nihon Keizai Shimbun" pointed out in a recent report that many countries and companies around the world are actively promoting the research and development of air batteries, hoping to achieve the goal of decarbonization as soon as possible.
Metal-air batteries have the advantages of good rate performance, high energy density, and low carbon sustainability. The famous scholar Leclanche developed the world's first metal-air battery in 1868. Today, a variety of metal-air batteries have been developed. Since the cathode reaction of most metal-air batteries is dominated by oxygen (in addition to carbon dioxide, nitrogen, etc.), the charge-discharge process is based on the oxygen reduction (ORR) and oxygen evolution (OER) reactions that occur in the cathode region.
Lithium-air battery
Li-air battery research can be traced back to 1976, first proposed by Littauer and Tsai. The negative electrode of the battery is metal lithium, the positive electrode is a composite material with a reasonable pore structure and ORR catalytic activity, the separator is glass fiber or PP film, and the electrolyte is generally 1M LiTFSI dissolved in TEGDME or DMSO. During discharge, the negative electrode lithium loses electrons and becomes Li+, which migrates to the positive electrode after crossing the separator. On the positive side, with the assistance of the catalyst, oxygen obtains external circuit electrons to generate ORR reaction to generate intermediate ions O2−, Li+ and O2− combine to form LiO2, and then undergo further electrochemical reduction or chemical reduction to generate the final discharge product Li2O2. During charging, Li2O2 undergoes an oxidation reaction to form LiO2-x, which is further decomposed into Li+ and O2, and Li+ migrates back to the negative electrode and regenerates metallic lithium.
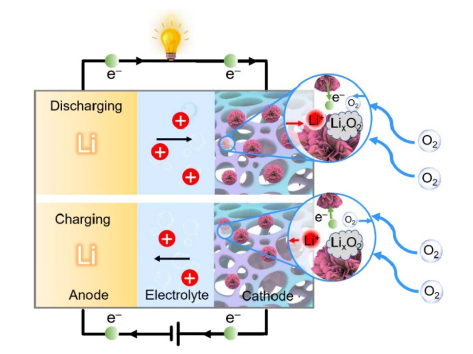
Oxygen is the real positive reactant throughout the battery's reaction process. As an important component of lithium-oxygen batteries, porous cathodes function to support active materials, provide a "gas-liquid-solid" three-phase interface for the electrochemical reaction between oxygen and lithium ions, and serve as ORR/OER during charge and discharge. catalyst for the process.
Aluminum-air battery
Metal aluminum was used in battery materials as early as the 19th century. In 1960, Zaromb et al. studied the theory of aluminum anode in air battery system in fuel cell, and discussed its feasibility. In 1962, Holzer F studied metal aluminum-air batteries experimentally. After that, after continuous development, in 1979, A.R.Despic et al. used seawater as the electrolyte for aluminum-air batteries and applied it in electric vehicles. Since 1990, aluminum-air batteries have been used in various fields, such as applications in chemical power sources, electric vehicles, and underwater submarines.
The negative electrode of the aluminum-air battery consumes metal aluminum and is composed of high-purity aluminum or aluminum alloy; the positive electrode consumes oxygen and is composed of a waterproof gas permeable membrane, a catalyst and a conductive material; the electrolyte consumes water and is composed of an alkaline solution or a saline solution. During the working process of the battery, metal aluminum dissolves in the electrolyte to form white metal hydroxide Al(OH)3, oxygen enters the positive electrode through the waterproof and breathable membrane, and electrochemically reacts with water under the action of the catalyst to generate OH-. Chemical energy is converted into electrical energy.
Zinc-air battery
The earliest research on zinc-air batteries can be traced back to 1879. Mache et al. used zinc flakes as the negative electrode of the battery. The air electrode was made of carbon and a small amount of Pt powder as the carrier, and the ammonium chloride aqueous solution was used as the electrolyte to assemble the earliest in history. zinc-air battery.
A zinc-air battery consists of a zinc anode, an electrolyte, an air cathode, and a diaphragm separator. The negative electrode can use pure zinc plate, zinc foil, zinc alloy, etc.; the electrolyte usually uses 6mol/L potassium hydroxide solution with good conductivity and O2 diffusion coefficient; the quality of the air positive electrode is the core of the zinc-air battery, Usually, a three-phase interface is formed by the gas diffusion layer in the gas phase, the catalyst layer in the solid phase and the current collector layer in the liquid phase. The membrane separator not only requires high sealing performance to avoid liquid leakage, but also ensures that the catalyst layer is in good contact with the air to facilitate the reaction.
During the discharge process of the zinc-air battery, the zinc of the negative electrode of the battery is oxidized in the electrolyte to form free zincate ions (Zn(OH)42-) and release electrons to the external circuit. (OH)42- ions gradually generate insoluble ZnO; at the three-phase interface of the positive electrode of the battery, O2 in the air accepts electrons from the negative electrode, and a reduction reaction occurs on the catalyst layer to become OH-, which is used to supplement the negative electrode consume OH- to maintain electrochemical equilibrium. The charging process is the reverse reaction process of discharge, that is, Zn(OH)42- gains electrons and is reduced to Zn at the anode, while at the cathode OH- loses electrons and desorbs O2.
Sodium-air battery
The sodium-air battery started late. In 2011, Peled et al. proposed that liquid molten sodium was used instead of metallic lithium as the anode to obtain a sodium-air battery that worked normally in the range of 105-110 °C. In recent years, research on sodium-air batteries has gained unprecedented momentum. The sodium-air battery is composed of a porous carbonaceous material as the air/oxygen cathode and pristine sodium metal as the anode. The two electrodes are sandwiched between a separator containing an aprotic electrolyte. The cathode reactant O2 diffuses from the surrounding air into the porous carbon during discharge.
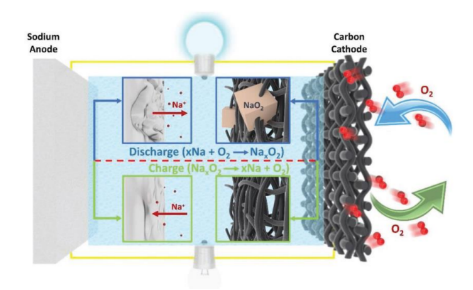
During discharge, metallic sodium is oxidized to sodium ions (Na+), which migrate to the cathode through the organic electrolyte. Simultaneously, O2 is reduced at the cathode surface to form reduced oxygen (O2- or O22-), which then combines with migrating Na+ to form metal oxides as discharge products. When the battery is fully charged, the reaction proceeds in the opposite direction, with Na-metal plating at the anode and oxygen escaping at the cathode.
Magnesium-air battery
Magnesium-air batteries generally use Mg and its alloys as the negative electrode, 6mol/L NaOH or KOH as the electrolyte, and the positive electrode is an air electrode, on which a mixture of catalyst and conductive carbon black is loaded. In the discharge work of the magnesium-air battery, the magnesium metal reacts with the electrolyte to release electrons, and the electrons reach the three-phase junction of the air electrode through the external circuit and react with oxygen and water to generate hydroxide ions.
At present, there are only a few experimental and theoretical reports on magnesium-air batteries, which are still in the early stage of development, and their cycle capacity is very limited.
Potassium-air battery
Potassium-air batteries realize the supply of electric current through a simple one-electron process without the use of expensive electrocatalysts. During discharge, it is easier to obtain thermodynamically stable KO2 due to the steric repulsion between K+, and KO2 is the only discharge product; KO2 is decomposed into K+ and O2 during the charging process, and the entire electrode reaction is realized by the single-electron redox couple O2/O2-.
Iron-air battery
The iron-air battery uses metallic iron as the anode, the air electrode as the negative electrode, and the alkaline or neutral salt solution as the electrolyte. In order to improve its activity, oxides or other elements are often added to the iron powder to improve the discharge capacity of the iron electrode.
Compared with other metal-air batteries, iron-air batteries have lower discharge voltage and specific energy density, and higher development and use costs, so there is less research on iron-air batteries at present. The most important factors affecting the electrode performance of iron-air batteries are the passivation of the iron electrode surface under high-rate discharge and the severe hydrogen evolution reaction inside the battery during the charging process. The hydrogen evolution reaction consumes about half of the battery energy and greatly reduces the Faradaic current efficiency.
Metal-air batteries use lightweight active metals as anode materials, which have the advantages of high energy density and stable discharge. In addition, its products are mainly metal oxides, which do not pollute the environment. It is an advanced energy technology with both energy and environmental protection. Currently, metal-air batteries have become the subject of intensive research worldwide and have made great strides in the past decade. It is expected to be used in new energy vehicles, portable equipment, stationary power generation devices and other fields in the future

